|
Video; Further info on Isoniazid induced Hepatotoxicity
|
Introduction
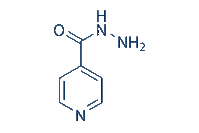
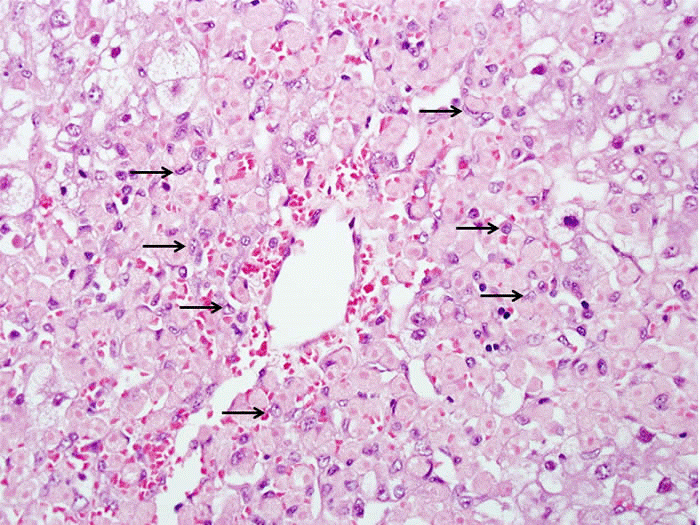
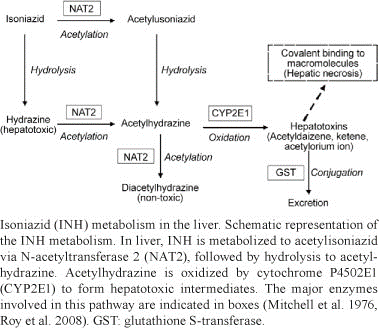
Isoniazid, also known isonicotinylhydrazine (INH), is an organic compound which is used as a first line medication, both prophylactically and in the treatment of tuberculosis (TB) or tubercle bacillus, an infectious disease caused by mycobacterial strains, usually mycobacterium tuberculosis. INH enters the mycobacterial cell by passive diffusion (1). INH is not toxic to the bacterial cell, but rather acts as a prodrug and is activated by a mycobacterial enzyme known as KatG (2). KatG is a multifunctional catalase-peroxidase which also has other activities including NADH oxidase and peroxynitritase (3,4). History (INH) was first discovered in the early 20th century but it wasnt until the 1950’s that its effective activity against TB was first realised (5). It was first tested on a Navajo community due to that particular reservation undergoing a very serious TB outbreak. However, as they had never previously been treated with Streptomycin, which was the main TB treatment at the time, this allowed for an accurate trial on the effectiveness of INH as a plausible treatment in the fight against TB (6). After the introduction of INH and later of rifampicin and ethambutol, TB became regarded as an easily treatable condition. However, due to new strains exhibiting multi drug resistance (MDR) and increased virulence, this is no longer the case (7) and as such, INH is still very much at the forefront in the treatment of TB. Side effects Side effects related to the use of Isoniazid include but are not limited to, mild CNS effects, rash, sideroblastic anaemia, peripheral neuropathy, hepatitis and generally abnormal liver function. Toxicity induced by INH is largely due to a single recessive gene responsible for drug acetylation rates. The speed at which a patient matabolises the drug will generally determine what type of toxic effects they are prone to. ‘Slow acetylators’ of the drug will be more likely to experience peripheral neuropathy caused by toxic levels of INH in the blood, however ‘fast acetylators’ of the drug will be prone to hepatotoxicity caused by the conversion of the acetylated drug to toxic intermediates (8). This is then converted to an acetylating agent that acts as a potent toxin, causing necrosis of the liver, however for many years this went unknown. INH-induced liver injury appears to be biochemically, clinically and histologically almost indistinguishable from viral hepatitis, except for the drug induced injury occurring primarily in people over 35 years of age (9). Earlier symptoms can include vomiting, nausea, loss of appetite and abdominal pain, followed later by dark urine and jaundice. Therefore patients must be monitored closely for any signs of INH induced hepatotoxicity during treatment. Liver injury - Mechanism of Action (MOA) Elevation of liver enzymes associated with liver injury, such as alanine transaminase (ALT), occur in approximately 10% -20% of INH recipients, with 1%-2% actually experiencing severe liver toxicity. However of the 1%-2%, only 0.01% will reach a stage of life threatening liver damage (10). This damage is caused by hepatic necrosis or death of liver cells, as Isoniazid is metabolized in the liver. The CYP450 family of enzymes play a key role in the metabolism of the drug (see figure 1). It first undergoes Phase I reactions through acetylation via the enzyme N-acetyltransferase 2 (NAT2) to form acetylisoniazid. This then undergoes hydrolysis to form acetylhydrazine (10). Acetylhydrazine is oxidised by a member of the cytochrome p450 family CYP2E1, to form hepatotoxic intermediates such as acetyldiazene, ketene and acetylorium ions. Glutathione S-transferase (GST) is involved in the phase II conjugation and excretion of the drug/toxic intermediates. With slow acetylators, the half life is between two and five hours, however with fast acetylators the half life is one to two hours, therefore leading to a build up of acetylisoniazid and toxic intermediates. These hepatotoxins are then available to bind covalently to macromolecules and liver proteins, causing necrosis of cells (see figure 2). INH also induces the P450 system and thus, acts as a source of free radicals (11). Free radicals are a source for oxidative stress and can act like a domino effect, thus leading to further liver damage. Figure 1. Metabolic processes leading to Isoniazid induced hepatotoxicity Figure 2. Sample of liver tissue, with arrows pointing to necrotic areas. Histologic Images Photomicrographs; Courtesy David E. Kleiner, MD, PhD, Laboratory of Pathology, National Cancer Institute.
Treatment of isoniazid induced hepatotoxicity As regards the treatment of INH induced hepatotoxicity, it presents a difficult challenge to manage due to a number of factors such as, patients affected are often on other potentially hepatotoxic drugs, making it more difficult to determine which is responsible for causing the damage. A lack of effective alternative drugs also means INH use is going to continue for the foreseeable future and patients who are severely affected by the drug may show few symptoms until potentially life threatening liver damage has already occurred. However in most cases toxicity is mild and can be resolved through stopping treatment for a period of time, allwing recuperation, although this may not always be an option. As such, necrosis of the liver through the use of drugs such as Isoniazid is always going to be a risk and is therefore something which must be factored into account in the treatment of TB cases. References
By; Ross McGeough |